Advisory
I advise medical imaging AI teams on technical decision-making, from early problem framing to R&D validation.
With a PhD in Computer Vision and over ten years of experience in applied medical imaging R&D, I bring structured perspective on problem formulation, data assumptions, experiment design, and technical feasibility throughout the R&D lifecycle.
My work ensures that technical choices, data constraints, and evaluation strategies remain aligned with real-world clinical and operational requirements, so that R&D efforts lead to robust and credible outcomes.
Engagements are scoped to project needs, from focused one-day assessments and technical reviews to longitudinal advisory mandates.
R&D Advisory Framework
A structured overview of how I intervene across the AI R&D lifecycle.
How I Work
I collaborate with MedTech teams through structured technical advisory, either via focused interventions or sustained involvement across project phases.
Some engagements address a specific question, such as feasibility assessment, data readiness, experiment design, or validation strategy, and can be completed in one to three days. Others involve longitudinal advisory support, contributing regular technical input as projects evolve.
The format adapts to project maturity and team structure, with the objective of strengthening judgment and technical defensibility throughout the R&D lifecycle.
Where I Intervene
My advisory work strengthens the AI R&D lifecycle from early framing to long-term technical defensibility.
1. Technical Framing & Feasibility
Clarifying objectives, constraints, problem formulation, and downstream implications before development decisions become locked in.
2. Data & Project Readiness
Identifying structural risks in datasets, infrastructure, documentation, and project setup that may affect development, validation, or deployment.
3. Experiment Design & Evaluation Strategy
Structuring experiments and evaluation protocols to ensure results are interpretable, comparable, and aligned with real-world use.
4. State-of-the-Art Review & Technical Positioning
Reviewing and selecting appropriate methods and approaches in alignment with project constraints and strategic objectives.
5. Ongoing Advisory & Due Diligence
Providing independent technical review of systems, documentation, or R&D programs to support internal decisions, partnerships, or investment discussions, with optional continued advisory follow-up.
Case Study
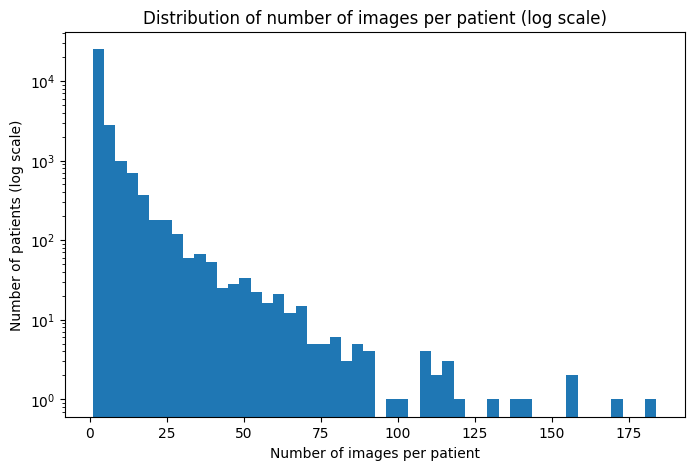
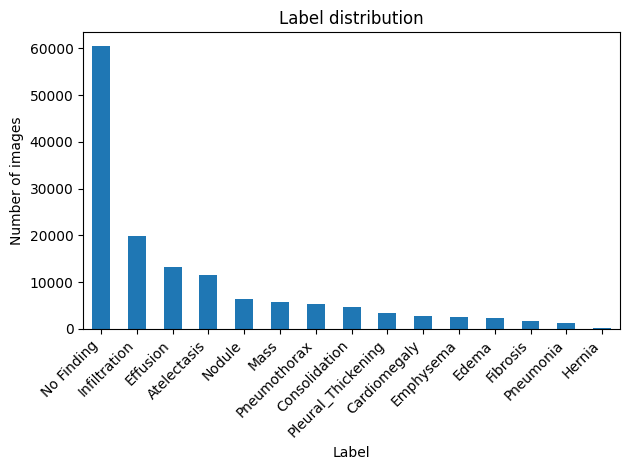
Project: NIH ChestX-ray14 Data Readiness Evaluation
|
Dataset Scale: 112,120 images / 30,805 patients |
|
Objective: Applying the VeraDP Data Readiness Protocol to identify the dataset’s strengths, limitations, and risks |
|
Identified Risks:
|
|
Strategic Outcome: The evaluation provides a framework for judgment under uncertainty, allowing R&D teams to account for data limitations before locking in technical and validation strategies. |
Who I Work With
- MedTech startups and scaleups developing AI-based products
- Healthcare and medical imaging teams advancing AI initiatives
- Technical and executive leaders seeking an independent sparring partner on AI R&D decisions
What This Enables
- Clearer technical positioning
- Stronger evaluation rigor
- Reduced hidden downstream risk
- Defensible performance claims
- Continuity of judgment over time
Why Work with Me
- Deep Technical Authority
PhD-level expertise in computer vision, combined with over ten years of applied MedTech R&D. - Experience at the R&D–Reality Interface
Hands-on involvement in projects where research constraints, data realities, and real-world clinical requirements must align. - Independent and Structured Perspective
An external technical reference bringing clarity and rigor to complex discussions. - Decision-Oriented Communication
Complex technical issues translated into structured, decision-ready insight for both technical and executive stakeholders. - International and Bilingual Context
Fluent in English and French, with experience across academic, clinical, and industrial environments.
Working on an AI project?
Let’s define a sound technical approach for your next decision.

